Beta particles are a form of ionising radiation resulting from radioactive decay. This article explores their nature, types, interactions, and applications. We will look into the historical discovery of beta particles, their physical characteristics, and their role in scientific and medical fields. Understanding beta particles is crucial in fields such as nuclear physics, radiology, and environmental science.
Introduction to Beta Particles
Beta particles are high-energy, high-speed electrons or positrons emitted by certain types of radioactive nuclei during a process known as beta decay. This form of radiation is part of the broader ionising radiation category, including alpha particles, gamma rays, and X-rays. Understanding beta particles requires a fundamental grasp of atomic structure and nuclear physics, as these particles are integral to the transformation processes occurring within unstable atomic nuclei.
Historical Context
The discovery of beta particles dates back to the early 20th century. Ernest Rutherford, a prominent physicist, identified beta particles as one of the three primary types of radiation emitted by radioactive substances, alongside alpha particles and gamma rays. His experiments with magnetic fields revealed that beta particles had a much smaller mass and higher velocity compared to alpha particles. This work laid the groundwork for modern nuclear physics and the study of radioactive decay processes.
Types of Beta Particles
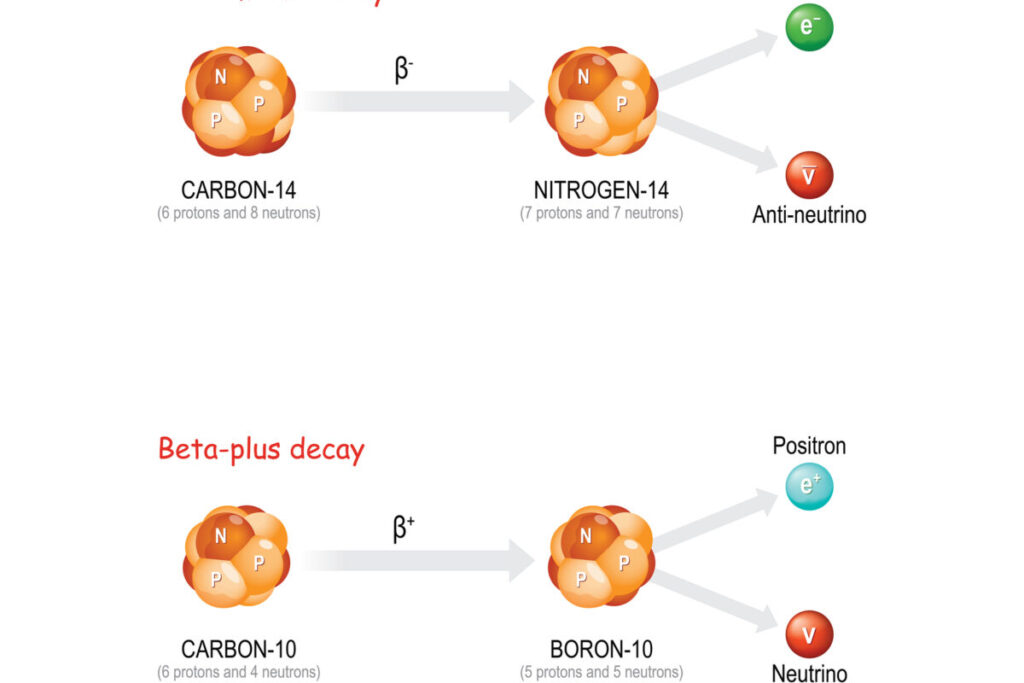
Beta particles can be classified into two distinct types based on their charge: beta-minus (β-) particles and beta-plus (β+) particles.
- Beta-Minus (β-) Particles are negatively charged electrons emitted from a neutron-rich nucleus. During beta-minus decay, a neutron is converted into a proton, an electron (the beta-minus particle), and an antineutrino. This process increases the atomic number of the element by one, forming a new element.
- Beta-Plus (β+) Particles: Also known as positrons, beta-plus particles are positively charged electrons. In beta-plus decay, a proton in the nucleus is transformed into a neutron, a positron (the beta-plus particle), and a neutrino. This process decreases the atomic number by one, also leading to the formation of a new element.
Physical Characteristics
Beta particles are characterised by their relatively small mass and high velocity. The mass of a beta particle is approximately 1/1836 that of a proton or neutron, which makes it significantly lighter. Their speed can approach a substantial fraction of the speed of light, giving them considerable kinetic energy. However, they have less ionising power compared to alpha particles but greater penetration ability.
A table summarising the key properties of beta particles:
| Property | Description |
| Type | Beta-Minus (β-) and Beta-Plus (β+) |
| Charge | β-: Negative (electron), β+: Positive (positron) |
| Mass | Approximately 1/1836 of a proton or neutron |
| Source | Emitted during beta decay from radioactive nuclei |
| Speed | Can approach a significant fraction of the speed of light |
| Penetration Ability | Moderate – can penetrate several millimetres of plastic or light metals, up to centimetres in tissue |
| Ionising Power | Less than alpha particles but more than gamma rays |
| Detection Methods | Geiger-Müller counters, scintillation counters, solid-state detectors |
| Medical Applications | Used in radiotherapy for treating cancer, diagnostic tracers |
| Industrial Applications | Thickness gauging, material analysis in the paper and plastic industries |
| Environmental Applications | Monitoring contamination, assessing safety of areas affected by nuclear activities |
| Safety Measures | Shielding with plastic, glass, or light metals; increasing distance; minimising exposure time; wearing protective clothing |
Interaction with Matter
When beta particles interact with matter, they ionise atoms by knocking out electrons from the outer shells of the atoms they encounter. This ionisation process can cause chemical changes in the material. Beta particles lose energy through collisions and interactions with electrons in the material, gradually slowing down until they are absorbed or come to rest.
The penetration ability of beta particles varies based on their energy. Low-energy beta particles can be stopped by a few millimetres of plastic or a sheet of paper. In contrast, high-energy beta particles may penetrate several millimetres of aluminium or other light metals. In biological tissues, beta particles can travel a few millimetres to a centimetre, causing ionisation along their path and potentially leading to biological damage.
Detection and Measurement
Detecting and measuring beta particles involves specialised equipment. Some common devices include:
- Geiger-Müller Counters: These are widely used for detecting beta radiation. The Geiger-Müller tube contains a gas that ionises when beta particles pass through, producing a detectable electrical pulse.
- Scintillation Counters: These devices use a scintillating material that emits light when struck by beta particles. The light is then converted into an electrical signal by a photomultiplier tube.
- Solid-State Detectors: These detectors use semiconducting materials to generate an electrical signal when beta particles interact with the detector material.
Applications of Beta Particles
Beta particles have numerous applications across various fields:
- Medical Uses: Beta particles are used in radiotherapy to treat certain types of cancer. Radioactive isotopes that emit beta particles, such as iodine-131, are used to target and destroy cancerous cells while minimising damage to surrounding healthy tissue.
- Industrial Applications: Beta particles are employed in thickness gauging and material analysis. For instance, they are used in the paper and plastic industries to ensure products have the correct thickness and density.
- Scientific Research: Beta particles play a crucial role in nuclear physics research. They are used to study nuclear reactions, decay processes, and the properties of various isotopes.
- Environmental Monitoring: Beta radiation monitors environmental contamination and assesses the safety of areas affected by nuclear activities.
Safety and Protection
Exposure to beta particles can be harmful, especially at high doses. Protection against beta radiation involves several strategies:
- Shielding: Materials like plastic, glass, or light metals can effectively shield against beta particles. The thickness of the shielding depends on the energy of the beta particles.
- Distance: Increasing the distance from the radiation source reduces exposure, as the intensity of radiation decreases with the square of the distance.
- Time: Minimising the time spent near a radiation source reduces the total exposure.
- Protective Clothing: Wearing protective clothing, such as lab coats, gloves, and safety glasses, helps reduce direct exposure to beta particles.
Future Perspectives
The study and application of beta particles continue to evolve. Advances in technology are leading to more precise detection methods and improved safety protocols. In medicine, new beta-emitting isotopes are being developed for targeted cancer therapies. In environmental science, beta particles are used to develop better monitoring and remediation techniques for contaminated sites.
Additionally, ongoing research in nuclear physics explores the fundamental properties of beta decay and its implications for understanding the universe. As we continue to refine our knowledge and technology, beta particles will remain a critical tool in scientific and industrial applications.
Conclusion
Beta particles, as a form of ionising radiation, play a significant role in numerous fields, from medicine to environmental science. Their unique properties and interactions with matter make them invaluable in both practical applications and fundamental research. By understanding the nature and behaviour of beta particles, we can harness their potential while ensuring safety and protection against their potential hazards. The future holds exciting possibilities for the continued utilisation and study of beta particles, promising advancements in technology, medicine, and our understanding of the atomic world.
Disclaimer
The content provided in this article, “Types of Beta Particles from Radioactive Decay,” is intended for informational and educational purposes only. It does not constitute professional advice in the fields of nuclear physics, radiology, health and safety, or any related discipline.
Readers should consult qualified professionals or institutions for advice specific to their situation, especially when working with radioactive materials or ionising radiation.
Use of radioactive substances must comply with relevant safety regulations and legal requirements in your country or region.